
在HSC化学模块6:酸/碱反应中挣扎?不要流汗!
In Year 12 Chemistry, acids and bases extend beyond flavour and litmus papers as we start looking at acids and bases at a much more molecular scale.
While there are many definitions of acids and bases (which you will learn later on in this module), acids are commonly known as proton donators while bases are proton acceptors.
These acids and bases are very important to our body, industry and environment, as much of life depends on pH, a measure of the concentration of protons produced by acids or consumed by bases.
In this article, we’ll be breaking down each of the inquiry questions in HSC Chemistry Module 6: Acid/Base reactions plus give you our top 3 tips on how to get a Band 6 in this module!
So, what are you waiting for? Let’s get to it!
Overview of Module 6: Acid/Base Reactions
How to get a Band 6 in HSC Chemistry Module 6
Overview of Module 6: Acid/Base Reactions
在我们深入研究每个询问问题之前,如果您需要回顾HSC化学的概念,make sure you head over to HSC Together here which has FREE video resources explaining concepts within each syllabus dot point!
询问问题1:什么是酸,什么是碱?
The first step to mastering anything is to learn the basics first! (No pun intended).
Here, we review what we know about acids and bases from junior high school. Acids are sour and bases are bitter. Easy right?
Remember, Year 12 extends junior high knowledge. While we merely scraped off balancing equations in junior high, it is now important to remember the products of each of the following reactions:
- Acid + Base → Salt + Water
- 酸 +碳酸盐→盐 +二氧化碳 +水
- Acid + Metal → Salt + Hydrogen
Neutralisation, the addition of acid and base together is particularly important, and you would need to revise Module 4: Drivers of Reaction to understand the thermodynamics of neutralisation.
提示:中和是放热的。你能解释为什么吗?
此外,酸和碱都有两个不同的定义。Arrhernius理论和Bronsted-Lowry理论。
| Arrhernius theory |
Bronsed-Lowry theory |
|---|---|
| - Acids ionize in water to form protons (H+) - Bases ionize in water to form hydroxide ions (OH-) |
- 酸是质子捐赠者 - Bases are proton acceptors |
It is important to know the definitions and theirlimitations准确地说,正如许多棘手的频段6个问题中,这将针对您的理解!
查询问题2:水在酸和碱的溶液中的作用是什么?
Acids and bases are good friends with water.
这是因为水允许浓缩。这些浓度将在确定pH和POH(分别衡量酸度和碱度)方面变得关键。
ph= -log10[H+]
poh = - log10 [哦 - ]
其中[]是摩尔/L中的浓度
ph+ pOH = 14
Where pH =1 is most acidic and pH = 14 is most basic.
重要的是要知道低pH或高pH值的含义。为了避免混乱,这是高pH值的含义以及低pH值的含义以及等等的摘要。
| ph | pOH | |
|---|---|---|
| 高的 | 低的concentration of H+ 低的acidity 高pH |
低的concentration of OH- 低碱度 低的ph |
| 低的 | 高浓度H+ 高酸度 低的ph |
高的concentration of OH- 高的basicity 高pH |
Yet, pH or pOH can be complicated depending on thestrengthof acids and bases
NB:Strength of acid/base is defined as its degree of dissociation.
Astrong酸/碱完全地dissociates in water while a虚弱的酸/碱does not完全地dissociate in water.
在弱酸/基地,这减少the concentration of H+ that increases the pH and decreases the concentration of pOH that decreases the pH.
尽量不要在酸/碱的强度和浓度之间混淆。浓缩或稀释的酸/碱与其强度无关。
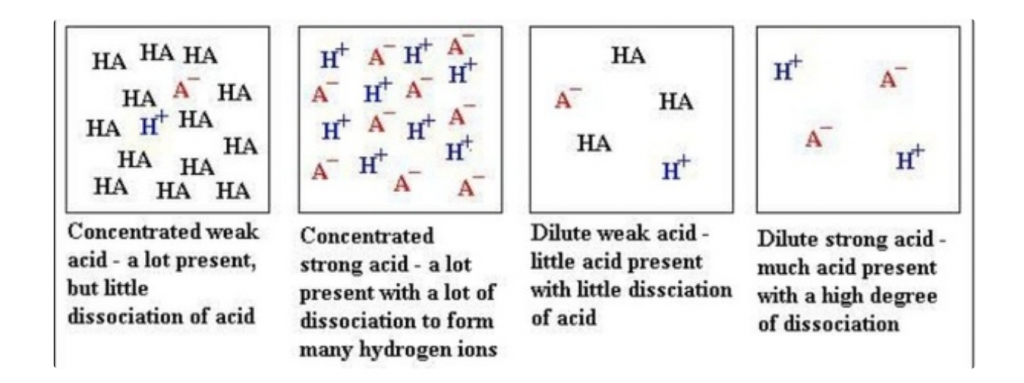
Sourced fromQuora
Inquiry Question 3: How are solutions of acids and bases analysed?
现在我们知道如何测量strongacids/bases, how do we do the same for虚弱的酸/碱?
Here is where titration comes in!
Titration is a technique that uses a solution of known concentration to measure the concentration of an unknown solution. This technique can be used for:
- Strong acids and strong base pairs
- Strong acids and weak base pairs
- 弱酸和强碱对
这些对有自己独特的滴定曲线和指示器!一定要记住他们!为了提供帮助,这是一个摘要:
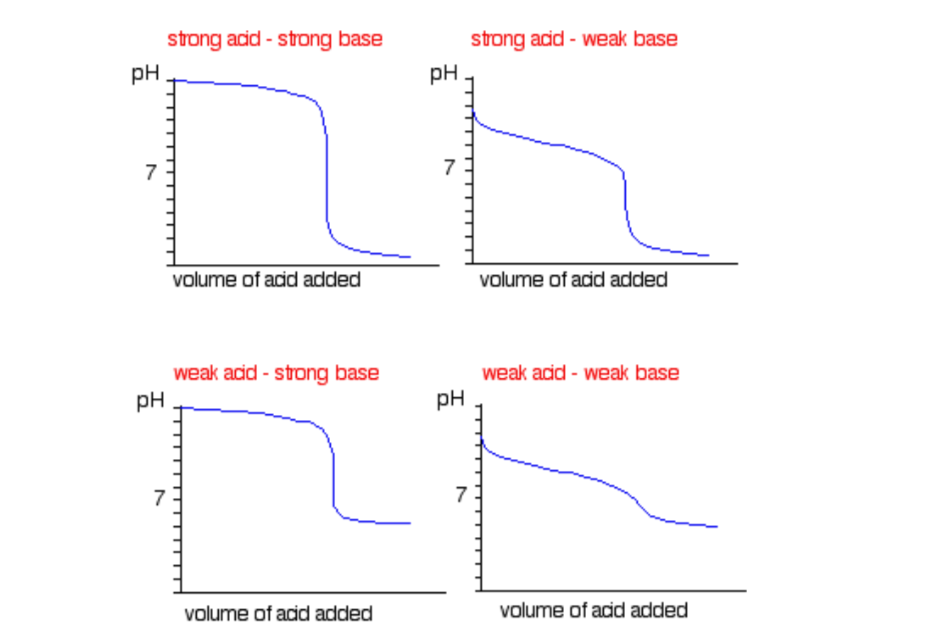
Sourced from化学指导
Note:弱酸和弱碱的滴定不需要12年级的水平,所以不要流汗!
| Titration pairs |
Best Indicators | Indicator end point pH range |
|---|---|---|
| Strong acid and strong base | 苯酚胺 | 8.3-10 |
| 强酸和弱碱 | Methyl Orange | 3.1-4.4 |
| 弱酸和强碱 | 苯酚胺 | 8.3-10 |
最重要的是,新的教学大纲引入了一种新公式,以定量测量强酸和弱酸之间的差异,即解离常数KA。
Note:最多虚弱的酸/碱反应是in equilibrium因为它们没有完全解离。同时,强酸/碱反应是not平衡,因为它们的反应完成了。
Ka is the measure of the dissociation extent of the acid. It is expressed as:
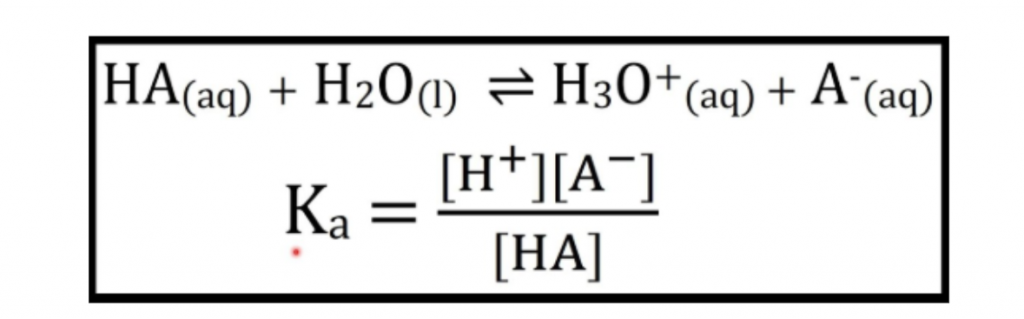
Ka looks very similar to Keq, and in fact, Ka ACTS like Keq too. It indicates whether the equilibrium reaction goes forward or backward.
ka越大,酸解离的越多,酸就越强。
Alternatively, the smaller the Ka, the less the acid dissociates, the weaker the acid.
如何在HSC化学模块中获得6条带6:酸/碱反应

提示#1:绘制酸和碱
Whew! Despite having only three inquiry questions, Module 6 does carry some heavy concepts!
为了组织您所知道的一切,请将它们编译成酸和碱基上集中的思维图!
Design your mind map such that each branch addresses each inquiry question.
这样,您可以从视觉上掌握一个概念与另一个概念的关系,形成一个可以用于修订的雨伞摘要!
For this module, be sure to include all necessary formulas as calculations play a pivotal role in acids and bases.
Tip #2: Master calculations
For examiners, Module 6: Acid/Base Reaction is a treasure box of calculation questions.
在滴定,pH,POH,PKA和稀释或浓缩溶液中有计算。
To prepare yourself for the trickiest of calculations, the best way is to练习,练习,练习。
这样,您不仅会形成一个例行程序来解决相同的样式问题,还可以使自己遇到更独特的问题,这些问题需要在框外面进行一些思考!
Lucky for you, these calculations have not changed much from the old syllabus’ “Acidic Environment”. Therefore, past HSC papers are an amazing resource!
Not enough time to complete papers? No worries.
工作簿,检查点化学是一个无所不包的资源,其中包括该主题最棘手的短而长的回答问题。
那你还在等什么?挖!
提示3:将理论应用于实践和现实生活中
Acid and base reactions occur all around us!
这就是为什么新的教学大纲要求您将酸/碱理论应用于工业,环境甚至生物学反应的原因。
请确保还将您在课堂上学到的理论应用于您所做的实践,尤其是为了滴定!
为了实践,请在修改酸和碱的实际应用时问自己这两个问题:
- 有助于其用途的酸或碱的特性是什么?
- How do the properties of acids and bases contribute to its uses?
通过问自己这些问题,您基本上是在掌握对化学的理解以及它们与现实生活的关系!
,结束我们的导游HSC化学模块e 6: Acid/Base Reactions! Good luck!
Need help in your other Chemistry modules? Check out our other guides below!
Looking for some extra help with HSC Chemistry?
We pride ourselves on our inspirational HSC Chemistry coaches and mentors!
We offer tutoring and mentoring for Years K-12 in a variety of subjects, with personalised lessons conducted one-on-one in your home or at our state of the art campus in Hornsby!
To find out more and get started with an inspirational tutor and mentorget in touch today!
Give us a ring on 1300 267 888, email us at[email protected]或检查我们Facebook!
Kate Lynn Law毕业于2017年,获得了全面的HSC奖,ATAR为97.65。她热衷于指导,她喜欢与高中生一起工beplay安卓西西软件作,以提高他们的学术,工作和生活技能,为HSC做准备以及接下来的事情。凯特(Kate)是一位狂热的博客作者,自2013年以来,自2013年以来就为2000多人管理了一个创意写作页面,自她十几岁以来就写信给国际观众。


